Introduction
What can you expect from BIOASTER?
We help you define and/or implement the technologies you need to develop your prophylactic, diagnostic, and therapeutic solutions in microbiology and infectious disease.
BIOASTER acknowledges that it requires a significant investment for you to identify, select, and access specialized technologies and experts with an extensive knowledge of microbiology and infectious disease.
When it comes to entrusting your projects, the de-risking of innovation, the ability to bring appropriate stakeholders together, quality, and a solid track record are the key determinants of a long-term relationship.
Furthermore, you may wish to overcome technological barriers and you are seeking partners who are willing to co-invest or share the risk associated. Thanks to our network, we are able to bring together appropriate stakeholders to help you achieve your goals.
Learn more about this, using an example (COVID-AuRA)
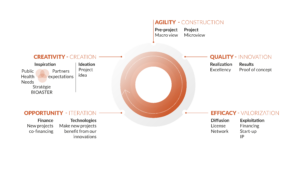
INNOVATING TOGETHER AND SHARING RISK IS IN OUR DNA; PROVIDING HIGH-IMPACT INNOVATION IS OUR MISSION
By its very nature as a Scientific Cooperation Foundation, BIOASTER proposes a risk management model for innovation from the design (or co-design) to the realization (or co-realization) stages, through action at three complementary levels:
- Sharing of scientific risks: BIOASTER contributes directly to your project through its own expertise and skills, as well as by assuming the management of the project. BIOASTER can also integrate the expertise of our network of academic and industrial partners to federate the most appropriate expertise and means.
- Sharing technological risks: Upstream or as a component of your project, BIOASTER helps you overcome technological barriers or brings together technological building blocks to create a model, a device, or a solution that can be deployed to facilitate your project. These technologies are developed and exploited within Technology Units, which as a whole constitute a unique technological value chain, spanning sampling to data analysis.
- Sharing financial risks: We aim to co-invest with you according to the nature of the project and its possible valorization (click here to see examples). Typically, we offer an intellectual property agreement that guarantees you full exploitation in the context of the products you develop and at the same time allows BIOASTER to exploit the technology in other fields.
You can find our detailed business model here.
CARE, MANAGEMENT, AND DELIVERY
We offer an end-to-end collaboration from conception to the creation of added value.
We first ensure correct understanding of your requirements and the expected deliverables, in order to design or co-design tailor-made projects or comprehensive programs.
We implement program or project management through a dedicated manager to ensure that constructive and regular discussion takes place. Together, we assess the possible risks or bottlenecks for the project and implement a mitigation plan. In addition, we specify key No/No go milestones to optimize the value obtained for your investment.
Our commitment is to provide you with robust, quality data that meet your industrial needs.
Beyond our own teams, we can access an extensive network of partners and experts in academic and private research that can provide valuable contributions to your program.
Whether it is a small or a large project, and throughout its life, our constant endeavor is to assist you to make well-informed and timely decisions.
A TEAM WITH A COLLABORATIVE MINDSET AND A RANGE OF COMPLEMENTARY EXPERIENCE AND SKILLS
We ensure that the full range of our partners’ cultures and needs are taken into account. We have a recruitment policy aimed at promoting a diversity of backgrounds, both academic or pharmaceutical and geographical. Indeed, more than 15 different countries are represented at BIOASTER.
Our diversity and values: agility, integrity, and sharing are our key values, and these permit us to better understand our partners and manage projects or programs, knowing that there are never two identical situations.
QUALITY TO SATISFY OUR PARTNERS
Your satisfaction is highly important to us. We are recognized by ISO 9001: 2015 quality management, which ensures that we can meet your needs at all the stages of a project, from design to realization. Our quality management process is underpinned by an approach that facilitates an approach of continuous improvement, through the assessment of performance across all of our activities.
All you need to know about our Quality Assurance policy is here.
A SOLID TRACK RECORD OF DELIVERING TECHNOLOGICAL SOLUTIONS
BIOASTER’s scientists can draw on experience in over 250 research programs undertaken with clients and partners. BIOASTER’s intellectual contributions to the innovation of their partners and clients are illustrated by the number of scientific publications generated, including peer-reviewed articles, book chapters, posters, presentations by key opinion leaders, and other publications.
Moreover, our scientists’ contributions have led to more than 20 patent applications to date. We have teamed up with and served more than 100 partners and clients, including pharmaceutical, biotech, diagnostic and life-science companies, as illustrated by a number of case studies.